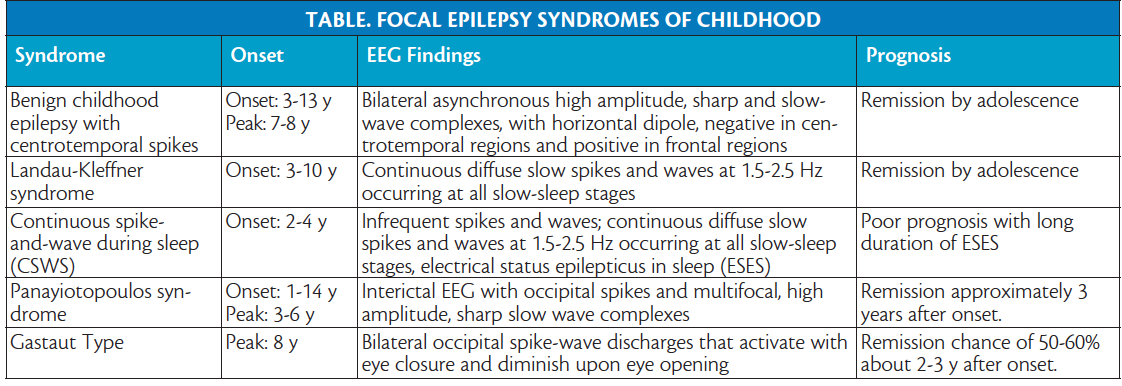
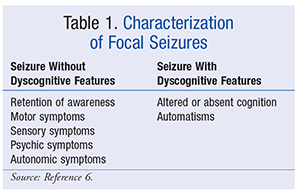
![PDF] Practice guideline update summary: Efficacy and tolerability of the new antiepileptic drugs I: Treatment of new-onset epilepsy | Semantic Scholar PDF] Practice guideline update summary: Efficacy and tolerability of the new antiepileptic drugs I: Treatment of new-onset epilepsy | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/aaa2aa01bedce63001c5b101b31c008efbb98bf2/3-Table1-1.png)
PDF] Practice guideline update summary: Efficacy and tolerability of the new antiepileptic drugs I: Treatment of new-onset epilepsy | Semantic Scholar

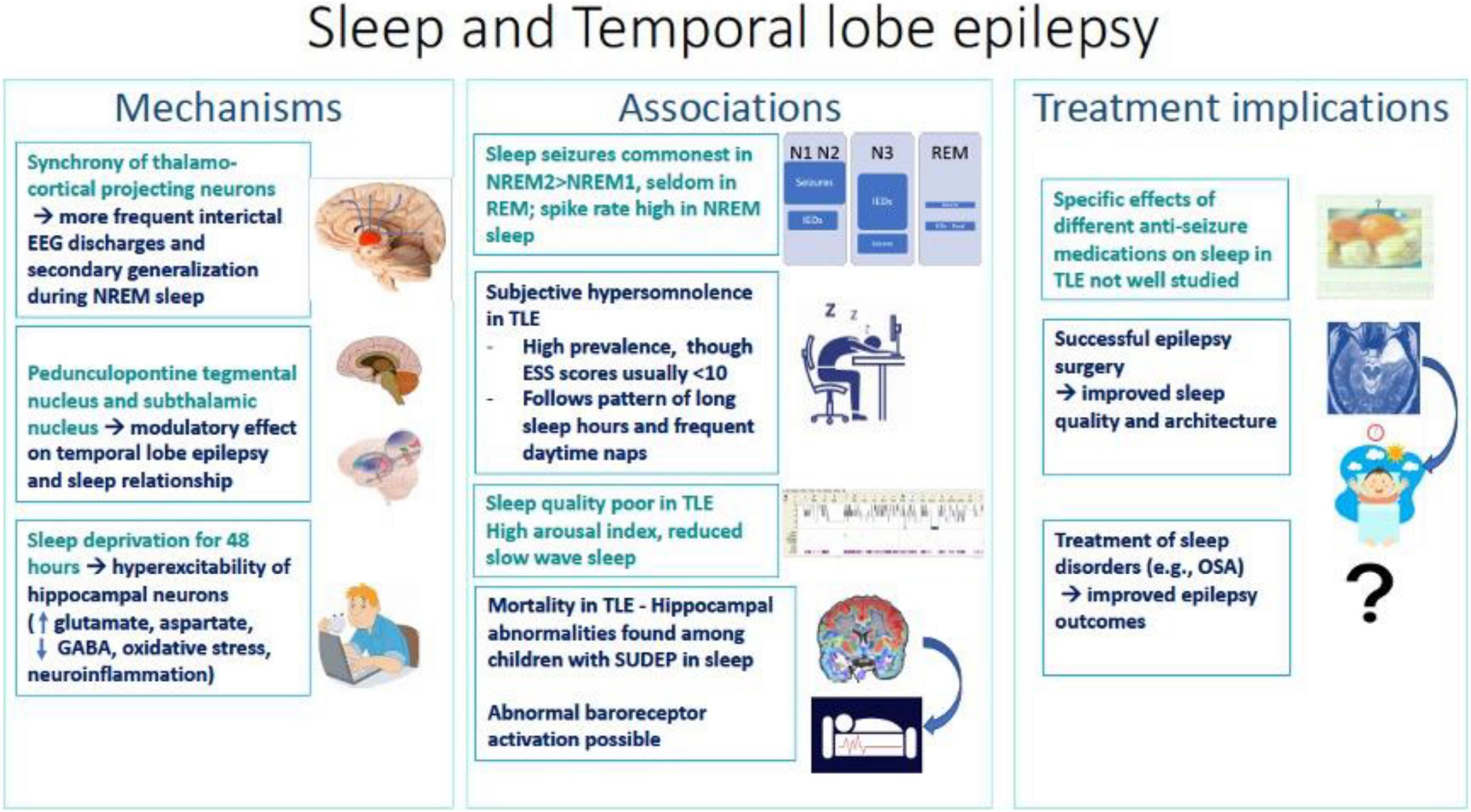
Focal abnormalities in idiopathic generalized epilepsy: A critical review of the literature - Seneviratne - 2014 - Epilepsia - Wiley Online Library

Characteristics of widely used third generation antiepileptic drugs... | Download Scientific Diagram

:max_bytes(150000):strip_icc()/rolandic-epilepsy-symptoms-causes-diagnosis-and-treatment-4689206-v1-1cc4661530af40c0a39dcb65bcc55e40.jpg)
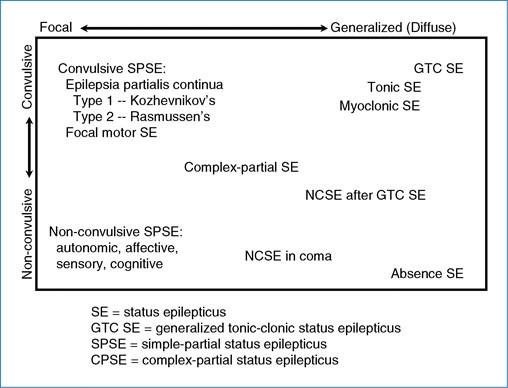



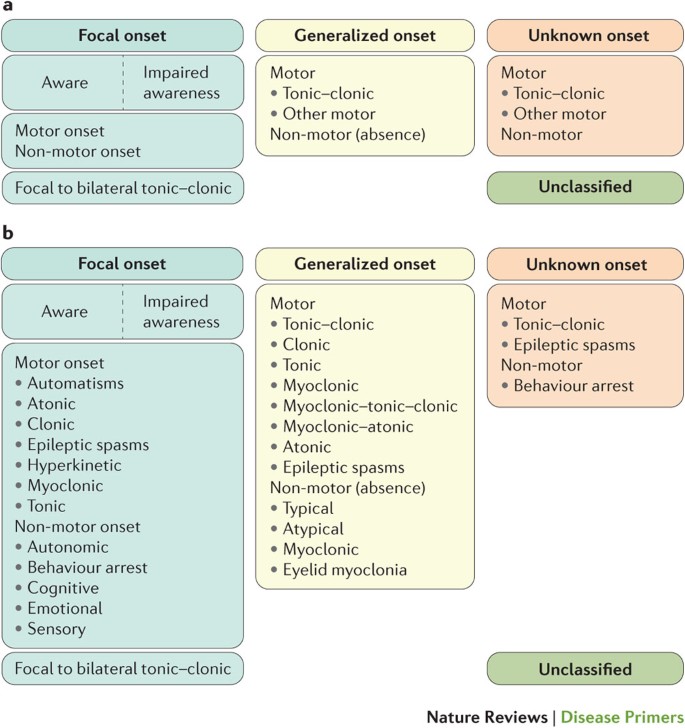




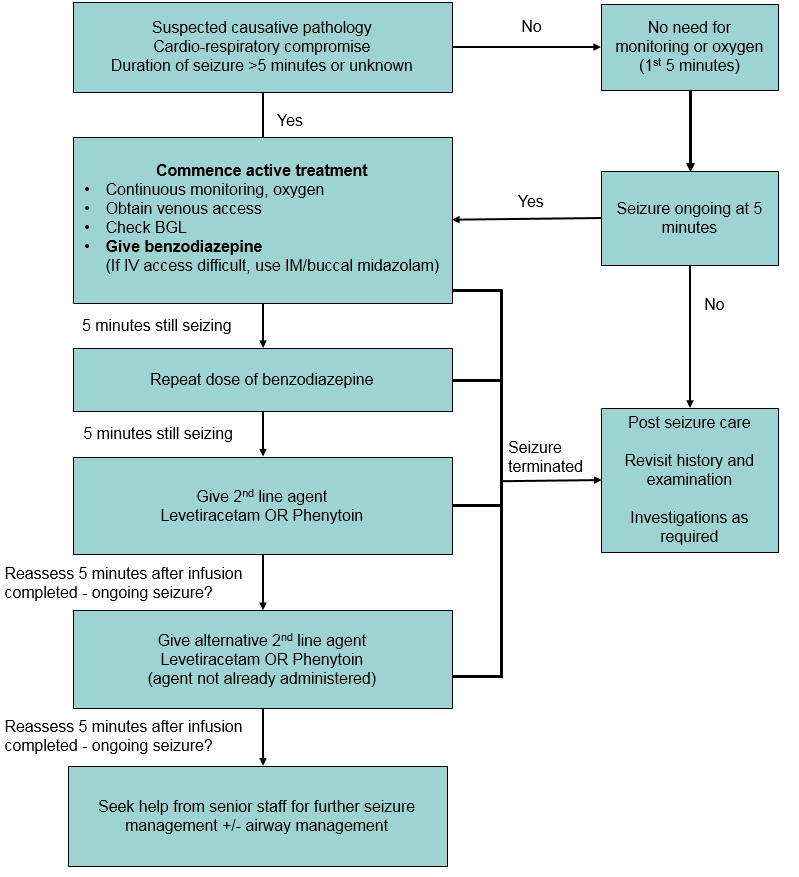
:max_bytes(150000):strip_icc()/adjunctive-epilepsy-treatment-5193083_final-01-5900f8be3105424089cc0d3858ba122c.jpg)